Dinitrogen tetraoxide, N2O4(g), decomposes to nitrogen dioxide, NO2(g), in a sealed syringe of volume 2 dm3.
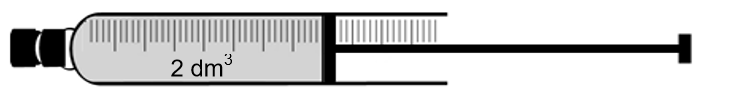
The mixture reaches equilibrium at 325 °C according to the following balanced equation:
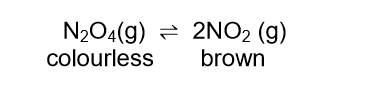
When equilibrium is reached, it is observed that the colour of the gas in the syringe is brown.
6.1 State Le Chatelier’s principle. (2)
6.2 The syringe is now dipped into a beaker of ice water. After a while the brown colour disappears.
Is the forward reaction EXOTHERMIC or ENDOTHERMIC? Explain the answer using Le Chatelier’s principle. (3)
6.3 The volume of the syringe is now decreased while the temperature is kept constant.
How will EACH of the following be affected? Choose from: INCREASES, DECREASES or REMAINS THE SAME.
6.3.1 The number of moles of N2O4(g) (1)
6.3.2 The value of the equilibrium constant (1)
6.3.3 The rate of the forward and reverse reactions (1)
6.3.4 Initially X moles of N2O4(g) were placed in the syringe of volume 2 dm3. When equilibrium was reached, it was found that 20% of the N2O4(g) had decomposed. If the equilibrium constant, Kc, for the reaction is 0,16 at 325 °C, calculate the value of X. (8)
[16]
