A test tube containing a straight chain organic acid X, ethanol and a catalyst is heated in a water bath as illustrated below.
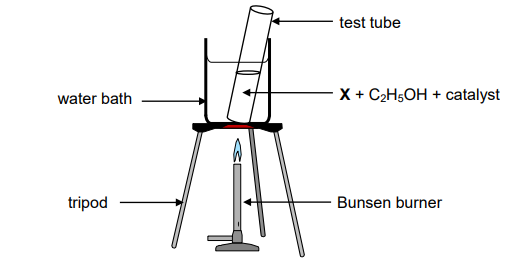
Organic compound Y is produced according to the following equation:
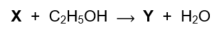
2.1 Give a reason why the test tube is heated in a water bath instead of directly
over the flame. (1)
2.2 Write down the:
2.2.1 Type of reaction that takes place here (1)
2.2.2 FORMULA of the catalyst needed (1)
2.2.3 Homologous series to which compound Y belongs (1)
The molecular mass of compound Y is 144 g∙mol-1 and its empirical formula is C4H8O.
2.3 Determine the molecular formula of compound Y. (2)
2.4 Write down the IUPAC name of compound Y. (2)
2.3 Write down the structural formula of the organic acid X. (2)
[10]
