An unknown gas, X2(g), is sealed in a container and allowed to form X3(g) at 300 °C. The reaction reaches equilibrium according to the following balanced equation:
3X2(g) ⇌ 2X3(g)
6.1 How will the rate of formation of X3(g) compare to the rate of formation of X2(g) at equilibrium? Write down only HIGHER THAN, LOWER THAN or EQUAL TO. (1)
The reaction mixture is analysed at regular time intervals. The results obtained are
shown in the table below.
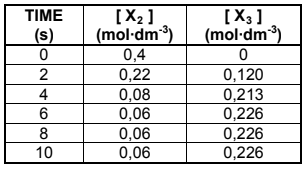
6.2 Calculate the equilibrium constant, Kc, for this reaction at 300 °C. (4)
6.3 More X3(g) is now added to the container.
6.3.1 How will this change affect the amount of X2(g)? Write down INCREASES, DECREASES or REMAINS THE SAME. (1)
6.3.2 Use Le Chatelier’s principle to explain the answer to QUESTION 6.3.1. (2)
The curves on the set of axes below (not drawn to scale) was obtained from the results in the table on page 10.
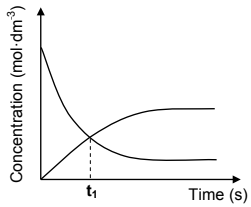
6.4 How does the rate of the forward reaction compare to that of the reverse reaction at t1? Write down only HIGHER THAN, LOWER THAN or EQUAL TO. (1)
The reaction is now repeated at a temperature of 400 °C. The curves indicated by the
dotted lines below were obtained at this temperature.
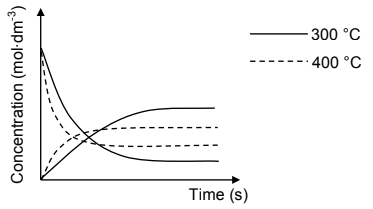
6.5 Is the forward reaction EXOTHERMIC or ENDOTHERMIC? Fully explain how you arrived at the answer. (4)
The Maxwell-Boltzmann distribution curve below represents the number of particles against kinetic energy at 300 °C.
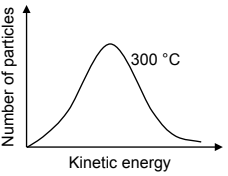
6.6 Redraw this curve in the ANSWER BOOK. On the same set of axes, sketch the curve that will be obtained at 400 °C. Clearly label the curves as 300 °C and 400 °C respectively. (2)
[15]
