Dilute acids, indicated in the table below, react with EXCESS zinc in each of the three experiments to produce hydrogen gas. The zinc is completely covered with the acid in each experiment.

The volume of hydrogen gas produced is measured in each experiment.
5.1 Name TWO essential apparatuses needed to determine the rate of hydrogen production. (2)
The graph below was obtained for Experiment 1.
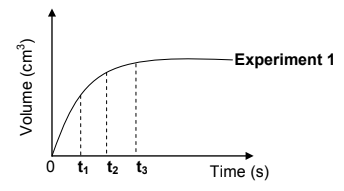
Use this graph and answer the questions that follow.
5.2 At which time (t1, t2 or t3) is the:
5.2.1 Reaction rate the highest (1)
5.2.2 Mass of zinc present in the flask the smallest (1)
5.3 In which time interval, between t1 and t2 OR between t2 and t3, does the largest volume of hydrogen gas form per second? (1)
5.4 Redraw the graph for Experiment 1 in the ANSWER BOOK.
On the same set of axes, sketch the graphs that will be obtained for Experiments 2 and 3. Clearly label the three graphs as EXPERIMENT 1, EXPERIMENT 2 and EXPERIMENT 3. (4)
5.5 The initial mass of zinc used in each experiment is 0,8 g. The balanced equation for the reaction in Experiment 3 is:
Zn(s) + 2HCl(aq) → ZnCl2(aq) + H2(g)
5.5.1 Calculate the mass of zinc present in the flask after completion of the reaction in Experiment 3. (5)
5.5.2 How will the mass of zinc present in the flask after completion of the reaction in Experiment 2 compare to the answer to QUESTION 5.5.1? Write down only LARGER THAN, SMALLER THAN or EQUAL TO. (1)
[15]
