In an experiment to investigate the relationship between pressure and temperature of
an enclosed gas, 48 g of oxygen gas was sealed in a container. The results obtained
are recorded in the table below.
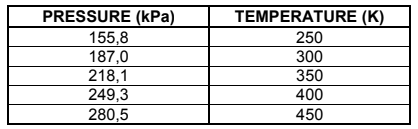
4.1 Draw a graph of pressure versus temperature on the attached ANSWER
SHEET. Extrapolate the graph so that it intersects the y-axis. (4)
4.2 What conclusion can be made from the final graph? (2)
4.3 Explain why it will not be possible to obtain accurate values at very low temperatures. (2)
4.4 Use the kinetic molecular theory to explain the effect of an increase in
temperature on the pressure of a gas. (4)
4.5 Under which conditions of temperature and pressure will a real gas act as an
ideal gas? (2)
4.6 Use the answer to QUESTION 4.6 to determine the volume of the container. (5)
[22]
