1.1 According to the Arrhenius theory, all bases …
A) are proton donors.
B) are proton acceptors.
C) form H3O+ ions in solution.
D) form OH‾ ions in solution. (2)
1.2 The number of neutrons in an atom of N157 is …
A) 5
B) 7
C) 8
D) 15 (2)
1.3 Which ONE of the following has the strongest forces between its molecules?
A) F2
B) Cℓ2
C) Br2
D) I2 (2)
1.4 Which ONE of the following has a tetrahedral shape?
A) + H3O
B) + NH4
C) CO2
D) AℓCℓ3 (2)
1.5 Two moles of H2 gas at STP occupy a volume of …
A) 2 dm3
B) 11,2 dm3
C) 22,4 dm3
D) 44,8 dm3 (2)
1.6 Which ONE of the following statements CORRECTLY describes the characteristics of an endothermic reaction?
A) ΔH is positive and the products have less potential energy than the reactants.
B) ΔH is positive and the products have more potential energy than the reactants.
C) ΔH is negative and the products have less potential energy than the reactants.
D) ΔH is negative and the products have more potential energy than the reactants. (2)
1.7 Which ONE of the following balanced equations represents a redox reaction?
A) H+(aq) + OH–(aq) → H2O(ℓ)
B) Mg(s) + CuSO4(aq) → Cu(s) + MgSO4(aq)
C) 2NaCℓ(aq) + Pb(NO3)2(aq) → 2NaNO3(aq) + PbCℓ2(s)
D) H2SO4(aq) + Ba(NO3)2(aq) → BaSO4(s) + 2HNO3(aq) (2)
1.8 During the extraction of gold, zinc powder is added to a solution of gold cyanide to produce gold according to the following balanced equation:
Zn(s) + 2NaAu(CN)2(aq) → 2Au(s) + Zn(CN)2(aq) + 2NaCN(aq)
The oxidising agent in this reaction is …
A) Au+
B) Zn
C) Na+
D) CN− (2)
1.9 The volume of a gas at a certain temperature and pressure is V.
If the temperature is doubled and the pressure is halved, the volume of the gas will be …
A) 4V
B) 2V
C) V
D) ½V (2)
1.10 Two identical containers, M and N, are shown below. Container M contains H2(g) and container N contains CO2(g). Both gases are at a temperature of 25 °C and a pressure of 101,3 kPa.
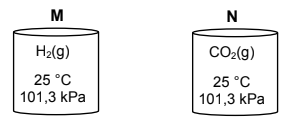
Consider the following:
(i) The average kinetic energy of the molecules is the same in both containers.
(ii) Container M contains more gas molecules than container N.
(iii) The mass of the gas in container N is greater than the mass of the gas in container M.
Which of the above statements is/are CORRECT?
A) (i) only
B) (iii) only
C) (i) and (ii) only
D) (i) and (iii) only (2)
[20]
