A barium hydroxide solution, Ba(OH)2(aq), reacts with a nitric acid solution, HNO3(aq), according to the following balanced equation:
Ba(OH)2(aq) + 2HNO3(aq) → Ba(NO3)2(aq) + 2H2O(ℓ)
The potential energy graph below shows the change in potential energy for this reaction.
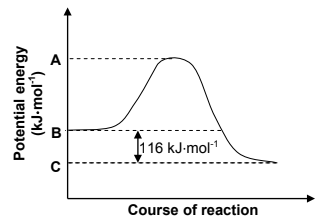
7.1 Is this reaction ENDOTHERMIC or EXOTHERMIC? Give a reason for the answer. (2)
7.2 Use energy values A, B and C indicated on the graph and write down an expression for each of the following:
7.2.1 The energy of the activated complex (1)
7.2.2 The activation energy for the forward reaction (1)
7.2.3 ∆H for the reverse reaction (1)
7.3 Calculate the amount of energy released during the reaction if 0,18 moles of Ba(OH)2(aq) reacts completely with the acid. (3)
[8]
