The simplified diagram below represents an electrolytic cell used to electroplate a
copper (Cu) coin with silver (Ag).
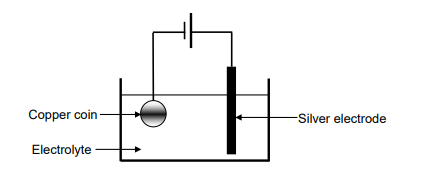
9.1 Define the term electrolysis. (2)
9.2 Which component in the diagram indicates that this is an electrolytic cell? (1)
9.3 Write down the NAME or FORMULA of the electrolyte. (1)
9.4 How will the concentration of the electrolyte change during electroplating?
Choose from INCREASES, DECREASES or REMAINS THE SAME.
Give a reason for the answer (2)
9.5 Write down the balanced equation of the half-reaction that takes place at the
silver electrode. (2)
[8]
