Learners are given the following two unknown half-cells:
Half-cell 1: Q2+ (aq) | Q(s)
Half-cell 2: Pt | R2(g) | R–(aq)
During an investigation to identify the two half-cells, the learners connect each half-cell alternately to a Cd2+(aq) | Cd(s) half-cell under standard conditions. For each combination of two half-cells, they write down the net cell reaction and measure the cell potential.
The results obtained for the two half-cell combinations are given in the table below.
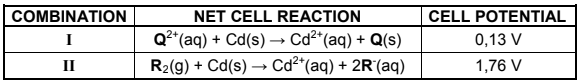
8.1 Write down THREE conditions needed for these cells to function as standard cells. (3)
8.2 For Combination I, identify:
8.2.1 The anode of the cell (1)
8.2.2 Q by using a calculation (5)
8.3 For Combination II, write down the:
8.3.1 Oxidation half-reaction (2)
8.3.2 NAME or FORMULA of the metal used in the cathode compartment (1)
8.4 Arrange the following species in order of INCREASING oxidising ability:
Q2+ ; R2 ; Cd2+
Explain fully how you arrived at the answer. A calculation is NOT required. (4)
[16]
