8.1 Consider the chemical reaction below:
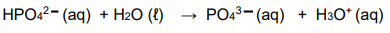
8.1.1 Define a base according to a Lowry-Bronsted theory. (2)
8.1.2 Write down ONE conjugate acid-base pair in the equation. (1)
8.1.3 Is the reaction mixture ACIDIC or ALKALINE at the completion of the
reaction? Give a reason for your answer. (2)
8.1.4 Write down the formula of a substance in the reaction, other than
H2O, that can act as an ampholyte in some reactions. (2)
8.2 Copper (II) oxide (CuO) reacts with nitric acid.
Write down a balanced equation for the reaction. (3)
8.3 40 g of IMPURE calcium carbonate reacts with a 200 cm3 of a dilute sulphuric acid with a concentration of 1,5 mol∙dm-3. All the calcium carbonate and sulphuric acid react completely leaving the impurities unreacted at the completion of the reaction.
8.3.1 Calculate the percentage purity of the calcium carbonate. (6)

To obtain the sulphuric acid solution of concentration 1,5 mol.dm-3 that
reacted with the IMPURE calcium carbonate, 10 cm3 of a concentrated
sulphuric acid solution of concentration 9 mol∙dm-3 was added to water.
8.3.2 Calculate the volume of water required to dilute the concentrated sulphuric acid solution to a concentration of 1,5 mol∙dm-3. (4)
[20]
