Learners use the reaction of MgCO3(s) with EXCESS dilute HCℓ(aq) to investigate the
relationship between temperature and the rate of a chemical reaction.
The balanced equation for the reaction is:
MgCO3(s) + 2HCℓ(aq) → MgCℓ2(aq) + CO2(g) + H2O(ℓ)
The results obtained are represented in the graph below.
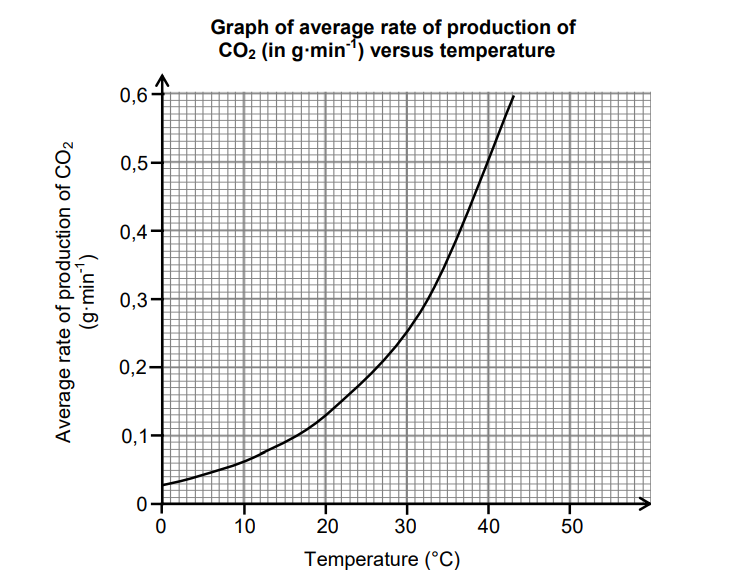
5.1 Define the term rate of reaction. (2)
5.2 State TWO conditions that must be kept constant during this investigation. (2)
5.3 Use the collision theory to explain the relationship shown in the graph. (4)
5.4 The learners obtained the graph above using 5 g MgCO3(s) with EXCESS
HCℓ at 40 °C.
Calculate the:
5.4.1 Time taken for the reaction to run to completion (6)
5.4.2 Molar gas volume at 40 °C if 1,5 dm3 CO2 is collected in a syringe (2)
5.5 The graph below represents the Maxwell-Boltzmann distribution curve for CO2(g) at 40 °C.
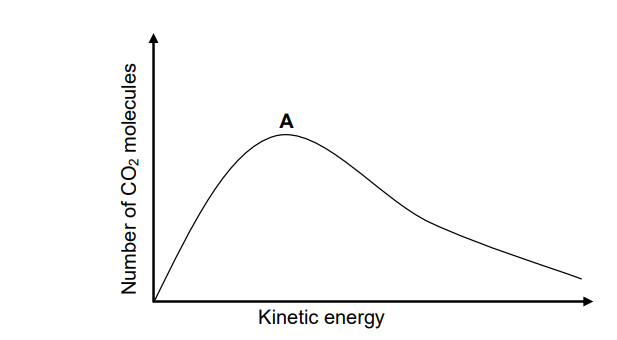
Redraw the graph above in the ANSWER BOOK. Clearly label the curve as A.
On the same set of axes, sketch the curve that will be obtained for the CO2(g)
at 20 °C. Label this curve as B. (2)
[18]
