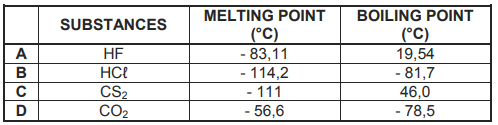
The melting points and boiling points of four substances (A, B, C and D) are shown in
the table below.
3.1 Define the term melting point. (2)
3.2 Explain the difference in melting points of HF and HCℓ by referring to the
TYPE of intermolecular forces. (4)
3.3 Which ONE of the substances (A, B, C or D) above is a liquid at 25 °C? (1)
3.4 Explain why CS2 has a higher boiling point than CO2. (3)
3.5 Which ONE of the substances (A, B, C or D) above has the highest vapour pressure? Give a reason for the answer by referring to the data in the table. (2)
[12]