Most elements found on Earth are metals. All metals have a metallic structure.
2.1 For a metallic structure, write down the:
2.1.1 Type of particles present (2)
2.1.2 Type of chemical bond formed between particles (1)
2.2 In which region on the periodic table are metals found? Choose from LEFT or
RIGHT. (1)
2.3 Write down the SYMBOL of a metal which is a liquid at room temperature. (1)
2.4 To prevent iron from rusting it is often mixed with other metals, e.g. stainless
steel is a mixture of iron, chromium and nickel.
2.4.1 Is stainless steel a HOMOGENEOUS or HETEROGENEOUS
mixture? Give a reason for the answer. (2)
2.4.2 Rusting is the formation of iron(III) oxide. Write down the FORMULA
of iron(III) oxide. (1)
2.5 The physical properties of two substances, A and B, are shown in the table
below.
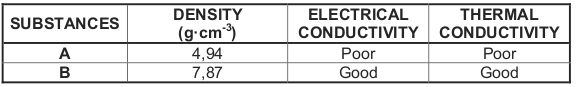
2.5.1 Which ONE of the substances, A or B, has the smaller mass to
volume ratio? Give a reason for the answer. (2)
2.5.2 Explain the difference between electrical conductivity and thermal
conductivity. (2)
2.5.3 Which ONE of the substances, A or B, is a metal? Give a reason for
the answer. (2)
[14]
