Learners prepare a solution of known concentration by dissolving 2 g pure sodium hydroxide crystals, NaOH, in water in a 250 cm3 volumetric flask.
7.1 Write down the term for the underlined phrase. (1)
7.2 Calculate the:
7.2.1 Concentration of the sodium hydroxide solution (4)
7.2.2 pH of the solution (4)
The learners now react 1,5 g of pure CaCO3 with 50 cm3 dilute HCℓ of unknown concentration. The EXCESS HCℓ is neutralised with 25 cm3 of the NaOH solution that they prepared. The balanced equations for the reactions are:
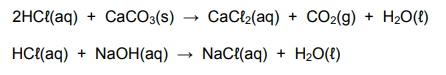
7.3 Calculate the initial concentration of the dilute HCℓ(aq). (8)
[17]
